With the development of science and technology, the emergence of new materials, and the improvement of technological levels, the market for empty capsules has gradually evolved from the original single-source animal gelatin capsules to a variety of plant-sourced chemically synthesized hydroxypropyl methylcellulose (HPMC) capsules, hydroxypropyl starch capsules, and naturally fermented pullulan polysaccharide capsules.

HPMC Empty Capsules (H-caps®)
Currently, in the empty capsule market, what are called vegetable capsules are hydroxypropyl methylcellulose empty capsules (hereafter referred to as HPMC capsules). Due to different formulation processes by various manufacturers, the products exhibit different disintegration behaviors in various media. The formulation process of HPMC capsules is introduced below.
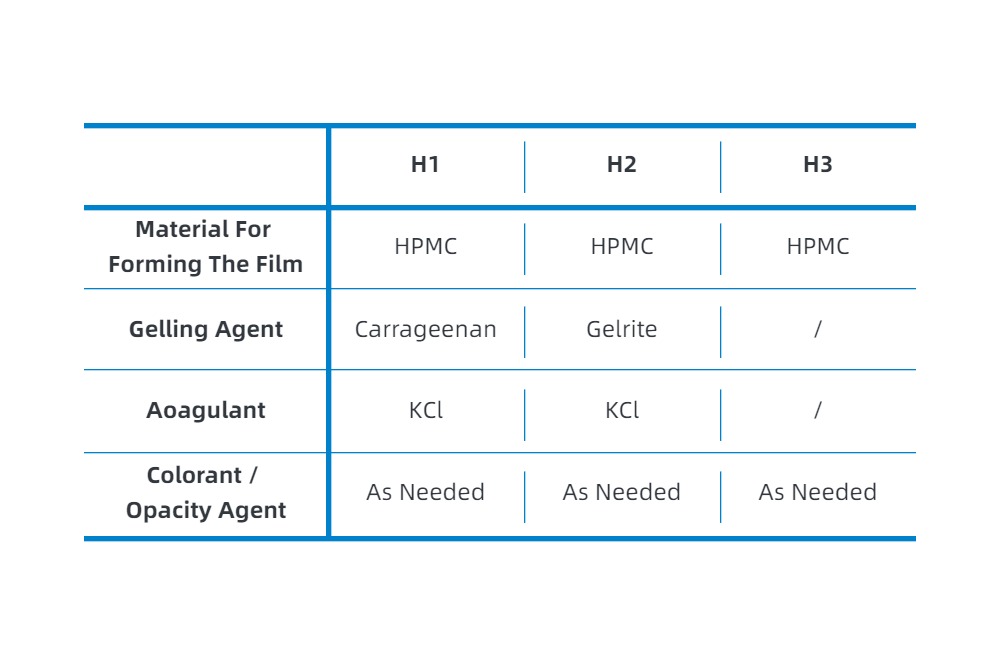
The water solubility and film-forming properties of hydroxypropyl methylcellulose, as well as its resistance to brittleness under low moisture conditions, make it a suitable main material for forming the film of empty capsules. Solutions of hydroxypropyl methylcellulose exhibit thermogelling properties, which do not show gel characteristics below the gelling temperature. This has led to the formulation process of HPMC capsules being divided into two categories: one with a gelling agent (cold gelling process) and one without a gelling agent (hot gelling process). In the cold gelling process, different choices of gelling agents are used, with the main market gelling agents being carrageenan and gelrite; different gelling agents and different gelling processes result in different disintegration/solubilization behaviors of HPMC capsules in different media. In the following text, products using carrageenan cold gelling process will be referred to as H1, those using gelrite cold gelling process as H2, and those without a gelling agent using hot gelling process as H3.
· Formulation Composition of Different HPMC Capsules
Only the main components are listed; some factories may add non-essential processing aids which are not listed here.
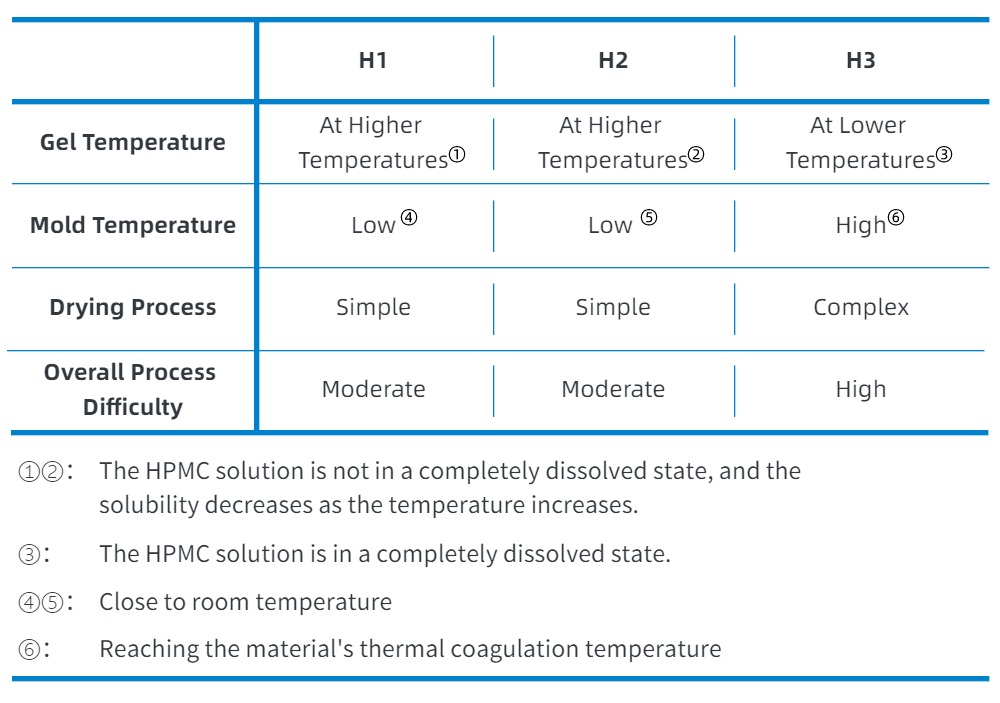
· Capsule Preparation Process
In the current empty capsule preparation process, the gelling nature of the gel solution is utilized, and the capsules are formed by dipping the molds into the gel solution, allowing the gel to form a film on the surface of the molds at a temperature difference between the gel solution and the mold.
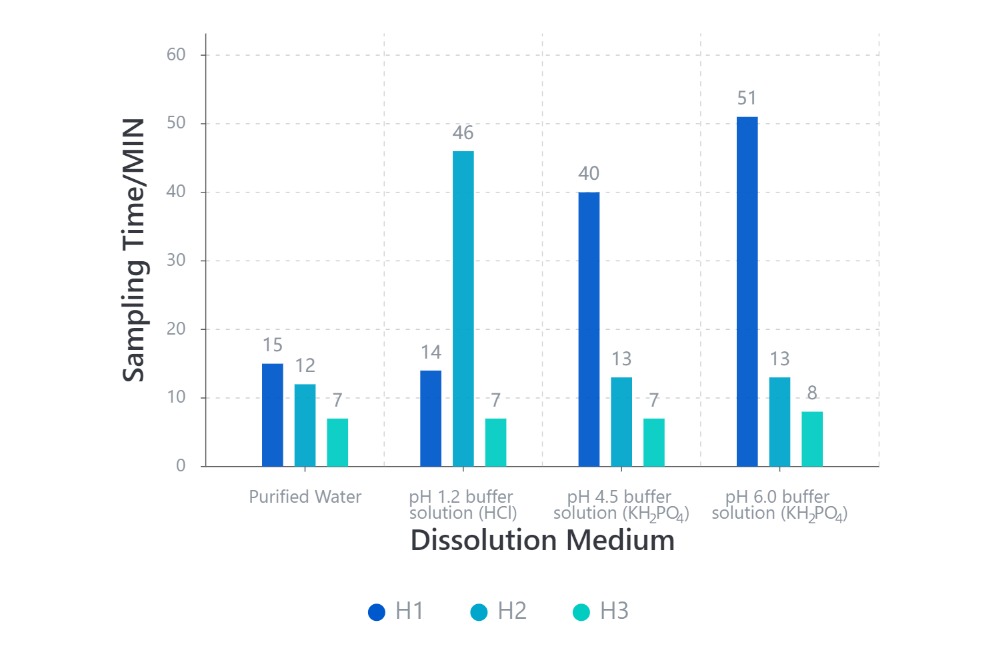
· Disintegration Performance of Different HPMC Capsules in Various Media
Samples are all #1 HPMC capsules.
· Solubilization Performance of Different HPMC Capsules in Various Media
Cefradine Capsules, 0.25g, paddle method
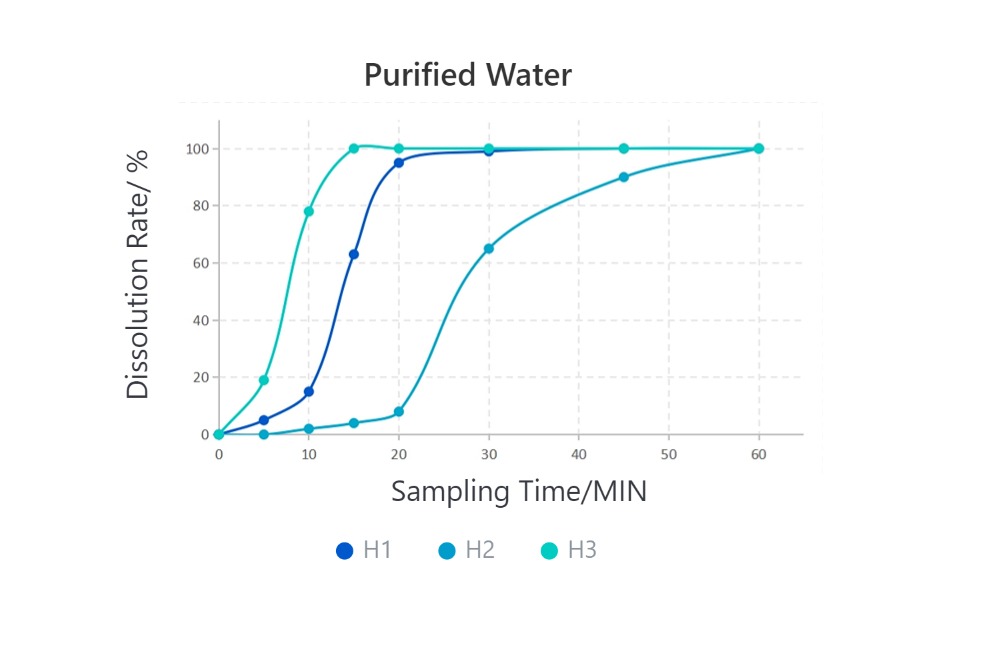
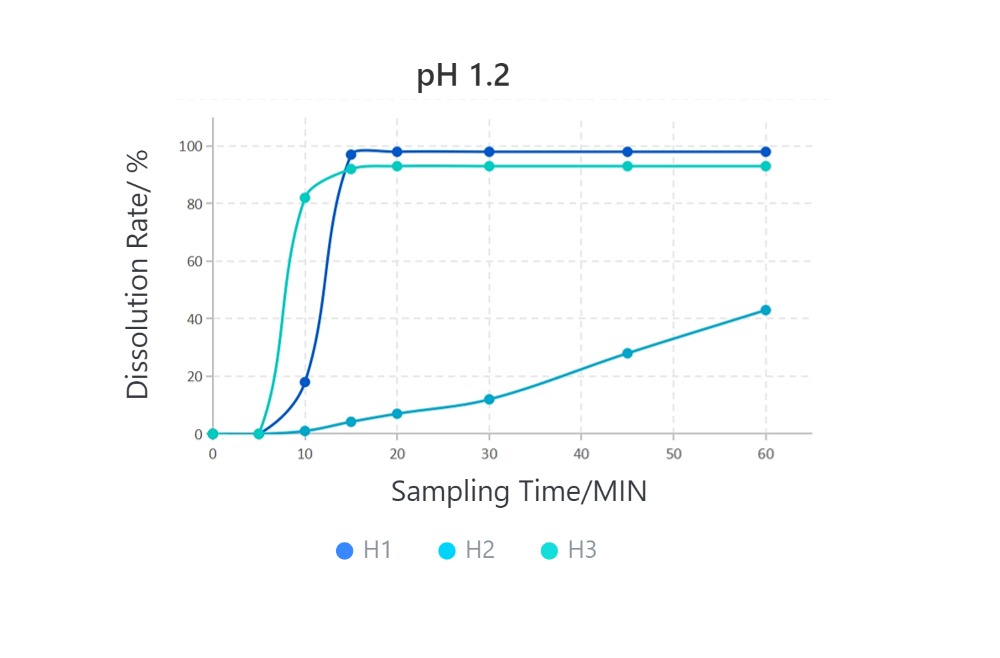
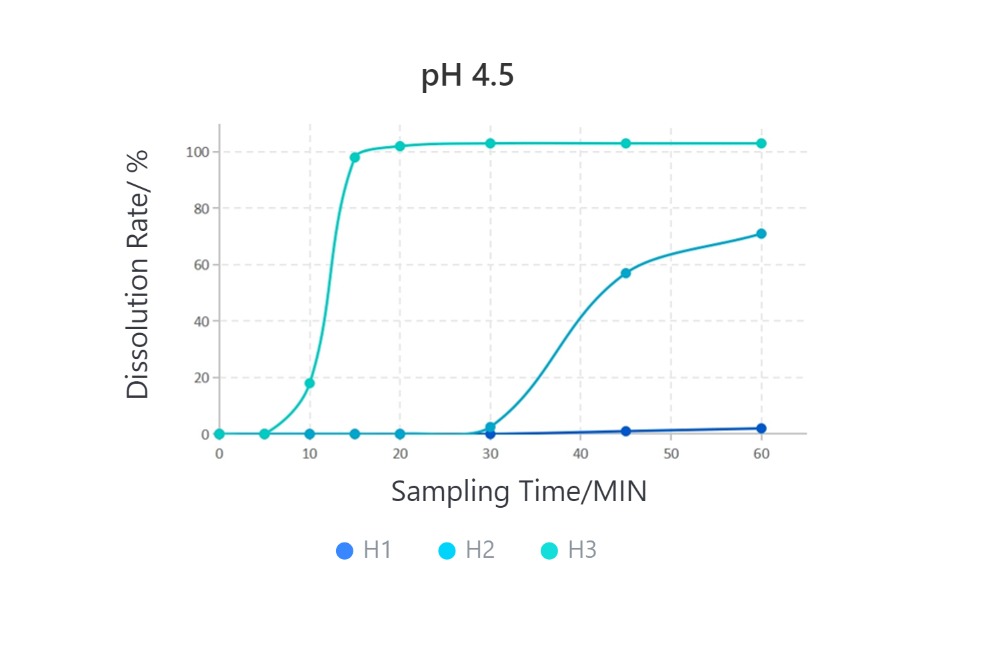
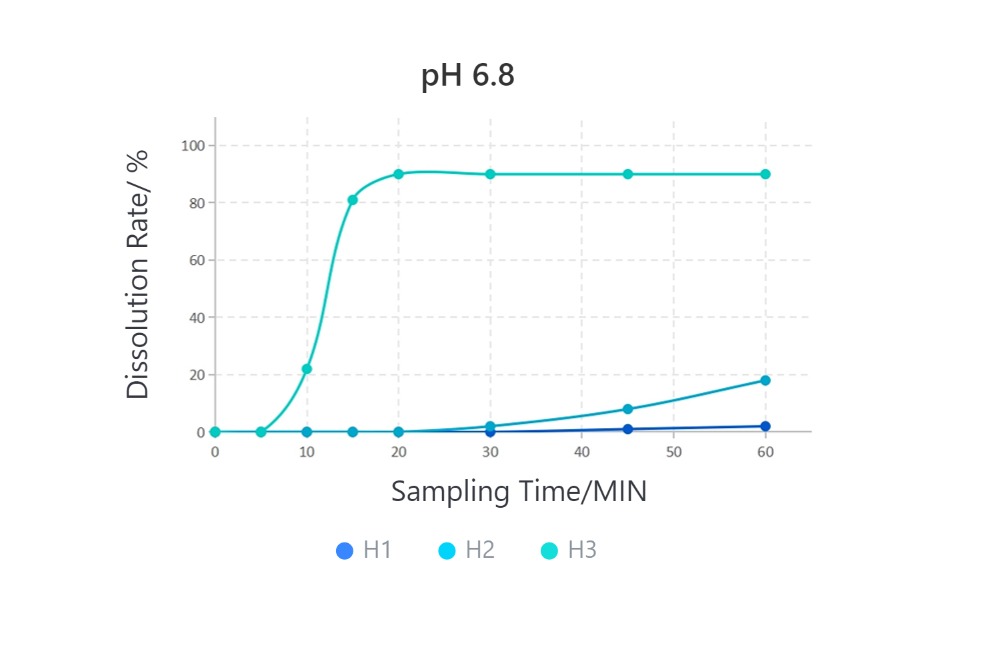
From the introduction above, due to their different formulation processes, the final empty capsule products exhibit significant variations in disintegration performance. In the application of capsule formulations, especially in medicinal gelling agents, the disintegration performance of empty capsules directly affects the solubility of the formulation. When you need to choose HPMC empty capsules, you should select the appropriate HPMC capsules based on the solubility requirements of the final formulation.